I am cycling my tank and getting all my water parameters right for the plants, then I will add stock. So currently no stock allowing me to mess around a bit and get things right. So I added way too much NH4 (ammonia and then NO2 and NO3 went through the roof, opps!! Its why I spend ages getting things right without stock).
I should add, I have a 100L tank and 120L RO storage tank. I dose the storage tank with GH and KH only before doing a water change.
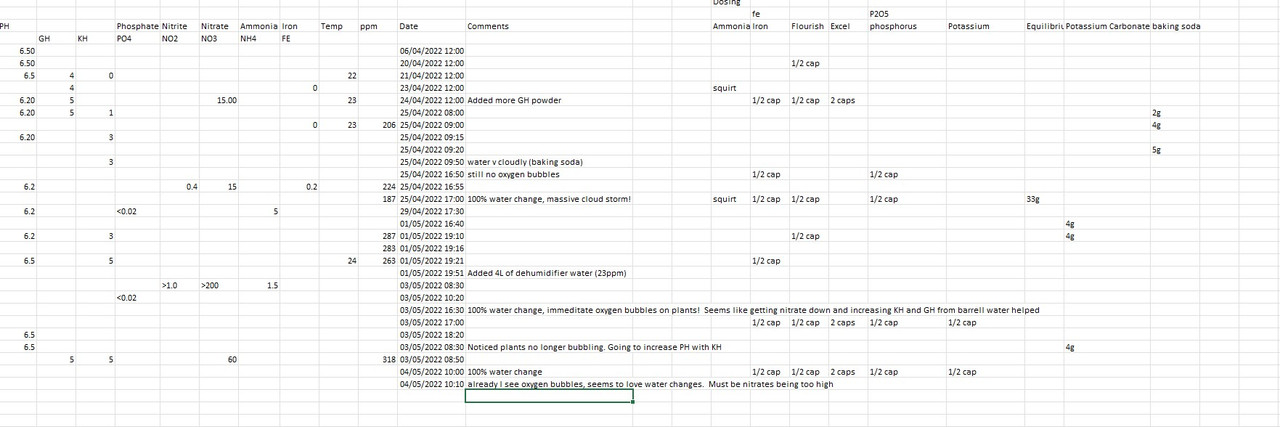
Noticed a weird thing, when I do a 100% water change, suddenly my plants start to pearl, ie lots of oxygen gets released (see pic below). Not sure why. The water the change I did has increased levels of KH and GH, and reduction of NO3 but that's about it. Once I do my water change I add micro nutrients as seen in the table below. But the plants produce loads of oxygen BEFORE I add micro nutrients so cant be that. The tank had a very high concentration of nitrate (I am adding NH4 manually as I cycle the tank and I added too much as mentioned above). See all my values below.
So wondered if it was related to either KH/GH change, NO3 reduction or something else. Below are all my values and also shows when the water was changed. I read that in general very high NO3 levels is okay for plants (of course lethal for fish and best to keep below 20mg/L). The first time it was over 200mg/L , when I did a water change the plants went mad, was lovely to watch. Then this morning I checked, and NO3 was still at 60mg/L so I did another 100% water change and once again the plants went mad!!
(water parameters of aquarium)
(water parameters of RO storage tank)

When I complete a 100% water change, the PH goes from 7.5 to 6.5 over time but I think that is the Co2 injection and the ADA v2 soil which buffers the water and lowers PH.
Thought maybe the plants wanted a higher PH, so when I added the new water (was 100% water change) they temporarily thrived in the more alkaline PH, then the CO2 and soil lowers it and once again the PH is 6.5 (ish) which could be too low and they stop pearling. The plants I am mainly referring to is Monte Carlo. Seems to go wild after water change!!
(Monte Carlo after water change)

I should also note I am using a drop checker to ensure I am injecting the correct amount of CO2.
Any help appreciated!



